The capabilities of OCT provide cardiologists with an effective tool for the assessment of patients with acute coronary syndrome (ACS), and permit new insights into plaque progression and ruptures. ACS is typically a life-threatening presentation of atherosclerosis. The in-depth lesion evaluation of OCT is invaluable and has enabled the pathophysiology of ACS to be fully unveiled.2
Acute Coronary Syndrome
Cardiovascular diseases are currently some of the leading causes of death and significant morbidities in developed countries. ACS encompasses three related but distinct clinical entities: unstable angina (UA), non-ST-segment elevation myocardial infarction (NSTEMI), and ST-segment elevation myocardial infarction (STEMI) (see Table 1). Management of ACS should encompass both acute and long-term strategies.
Pathophysiology of Acute Coronary Syndrome
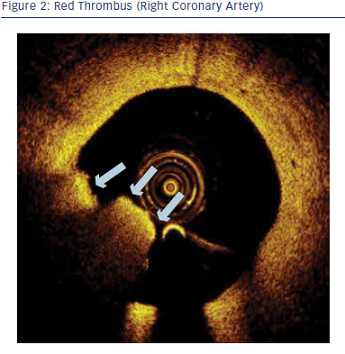
The pathophysiology of ACS is a dynamic process involving acute thrombosis leading to sudden luminal narrowing with an abrupt and often critical reduction in blood flow in the culprit coronary vessel. Plaque disruption and underlying inflammation were demonstrated to be the key causative factors. Three common plaque characteristics in ACS have been identified from autopsy studies, namely:
- plaque rupture (55–60 %);
- plaque erosion (30–35 %); and
- superficial calcified nodule (2–7 %).3
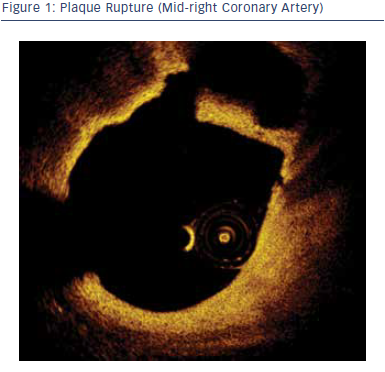
Plaque rupture can be characterised by the presence of fibrous cap discontinuity and a cavity within the plaque.4
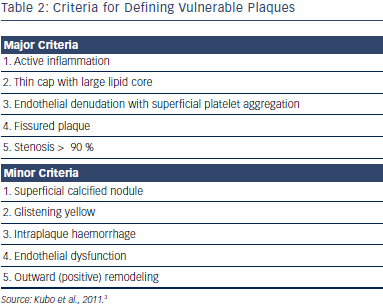
Plaque erosion can be identified by a thick intima lacking surface endothelium. The lipid core can be absent or small. A calcified nodule is typically defined as a protrusion that is signal-poor or as a heterogeneous region with a distinctively sharp border. Table 2 summarises the features of thrombosis-prone vulnerable plaques. OCT can readily identify most of these features.5
Optical Coherence Tomography Assessment of Culprit Lesions in Acute Coronary Syndrome
OCT is a promising tool to assess patients with ACS because it can detect plaques at high risk for rupture and provide additional information about plaque composition, thickness of the fibrous cap, the presence of thrombi and collagen composition. TCFA was the first lesion morphology studied with OCT in patients presenting with ACS.6 TCFA is defined as a signal-poor region comprised of rich lipid pool covered by a thin fibrous cap measuring < 65 μm. TCFA has been found more frequently in patients presenting with ACS than with stable angina (ACS 72 % versus stable angina 20 %).4
Kubo et al. demonstrated that OCT detection of plaque rupture, erosion and thrombus in acute myocardial infarction patients was comparable to that of previous histological studies.3 OCT had also been used to demonstrate different plaque characteristics, including plaque shoulder, fibrous-cap inflammation, ruptured plaque cavity size and direction of aperture opening in various presentations of ACS.7
In addition to assessment of plaque characteristics in ACS, OCT can also assist in prediction of outcome after percutaneous coronary intervention (PCI). Tanaka et al. reported a higher incidence of TCFA in the target lesions of ACS patients who suffered from no-reflow after PCI compared with patients with good reflow (50 % versus 16 %, p=0.005).4 Kato et al., on the other hand, demonstrated an increase in plaque vulnerability in the non-culprit lesions in patients with ACS compared with non-ACS patients, as evidenced by a larger lipid volume, thinner fibrous cap and higher prevalence of TCFA.8 This could explain the higher incidence of recurrent ischaemic events in ACS patients in the long term. OCT can also be utilised for assessment of ACS secondary to stent thrombosis. Studies have shown that the presence of neoatherosclerosis and plaque rupture in the neointima of previously stented segments, might contribute to the occurrence of very late stent thrombosis.9
In summary, OCT can be applied in the diagnosis and treatment of ACS for the accurate and detailed evaluation of plaque, the prediction of clinical outcome post-PCI and the assessment of late stent thrombosis.
Limitations
At present, OCT can provide high-quality and detailed assessment of plaque characteristics in ACS, which may influence peri-procedural decision-making during PCI as well as post-procedural optimisation. However, the translation of such advantages into clinical benefit requires further clinical research.
Conclusion
OCT is currently the state-of-the-art intracoronary imaging modality and can provide histology-grade assessment of the culprit lesion in ACS. OCT enables the detailed assessment and understanding of the pathophysiology of ACS that can be used to guide clinical decisionmaking during PCI enabling a more patient- and lesion-oriented approach. Ultimately, patient outcomes may be further enhanced. OCT will continue to play a pivotal role in the clinical management of ACS.
Special thanks to Drs. Arthur Yung and Anthony Wong for their assistance in the creation of this white paper.